Introduction to Living Matter Molecules
Living matter molecules, known as biomolecules, are the fundamental building blocks of life. They are primarily composed of carbon, hydrogen, oxygen, nitrogen, sulfur, and phosphorus, forming the basis of cellular structures and processes. Understanding these molecules is crucial, as they play essential roles in biological systems, from energy production to genetic information storage and transfer.
Biomolecules can be classified into four main categories: carbohydrates, proteins, lipids, and nucleic acids. Carbohydrates serve as energy sources and structural components. Proteins, made up of amino acids, are involved in virtually every cellular function, including catalyzing biochemical reactions and providing structural support. Lipids are vital for forming cell membranes and storing energy. Nucleic acids, such as DNA and RNA, are responsible for storing and transmitting genetic information, guiding cellular development and functioning.
The significance of living matter molecules extends beyond mere structural roles. These biomolecules interact intricately with one another, enabling complex biochemical pathways that sustain life. For example, enzymes, which are specialized proteins, catalyze biochemical reactions by lowering activation energy, allowing metabolic processes to occur efficiently. Moreover, the dynamics of molecular interactions inform various cellular functions, such as signal transduction and gene expression regulation.
A solid understanding of molecular biology is essential for exploring how living matter molecules influence life on Earth. Concepts such as molecular structure, function, and interactions not only form the foundation for advanced biological sciences but also pave the way for biotechnological applications. As research progresses, the insights gained from studying these molecules will continue to advance our knowledge of life itself and its complex interplay with the environment.
Understanding Atoms and Elements
Atoms and elements form the fundamental building blocks of all matter, including the living organisms that populate our planet. An atom is the smallest unit of an element, composed of a nucleus containing protons and neutrons, surrounded by electrons in defined energy levels. Elements are pure substances that consist of only one type of atom; they are organized in the periodic table based on their atomic number, which indicates the number of protons in an atom’s nucleus. Each element possesses unique properties that contribute to its behavior in chemical reactions and interactions.
Among the 118 known elements, four are particularly crucial for life: carbon (C), hydrogen (H), oxygen (O), and nitrogen (N), often referred to as the primary biological elements. Carbon is the backbone of organic molecules, possessing the unique ability to form stable bonds with other carbon atoms and various elements. This property allows for a vast array of complex organic compounds essential for life, such as carbohydrates, proteins, and lipids.
Hydrogen, the most abundant element in the universe, is important due to its role in forming water, a vital solvent that facilitates various biochemical processes. Oxygen, on the other hand, is essential for cellular respiration and energy production in living organisms, while nitrogen is a critical component of amino acids and nucleotides, which make up proteins and DNA, respectively.
Each of these elements has specific chemical properties that contribute to their biological roles. For instance, the ability of carbon to form multiple bonds results in diverse molecular structures, while the electronegativity of oxygen allows it to attract electrons, leading to the formation of polar molecules. Understanding the significance of these elements is crucial in comprehending the chemistry of life and how complex biological molecules arise from simple atomic interactions.
The Basics of Chemical Bonds
Chemical bonds are fundamental interactions that allow atoms to connect and form molecules, which are essential for the existence of living matter. The primary types of chemical bonds include ionic bonds, covalent bonds, and hydrogen bonds. Each type of bond varies in its nature of interaction and strength, playing a crucial role in the structure and function of biological molecules.
Ionic bonds arise when atoms transfer electrons from one to another, resulting in the formation of positively and negatively charged ions. These oppositely charged ions attract each other, leading to the creation of a stable ionic compound. In living organisms, ionic bonds are vital for the formation of key biological molecules, such as salts. For instance, sodium ions and chloride ions combine to form sodium chloride, an essential component of bodily fluids.
Covalent bonds, on the other hand, involve the sharing of electrons between atoms, which allows both participants to attain a stable electronic configuration. This type of bond is crucial for building organic molecules, such as carbohydrates, proteins, and nucleic acids. The strength and stability of covalent bonds enable these molecules to play central roles in processes such as energy transfer and genetic information storage.
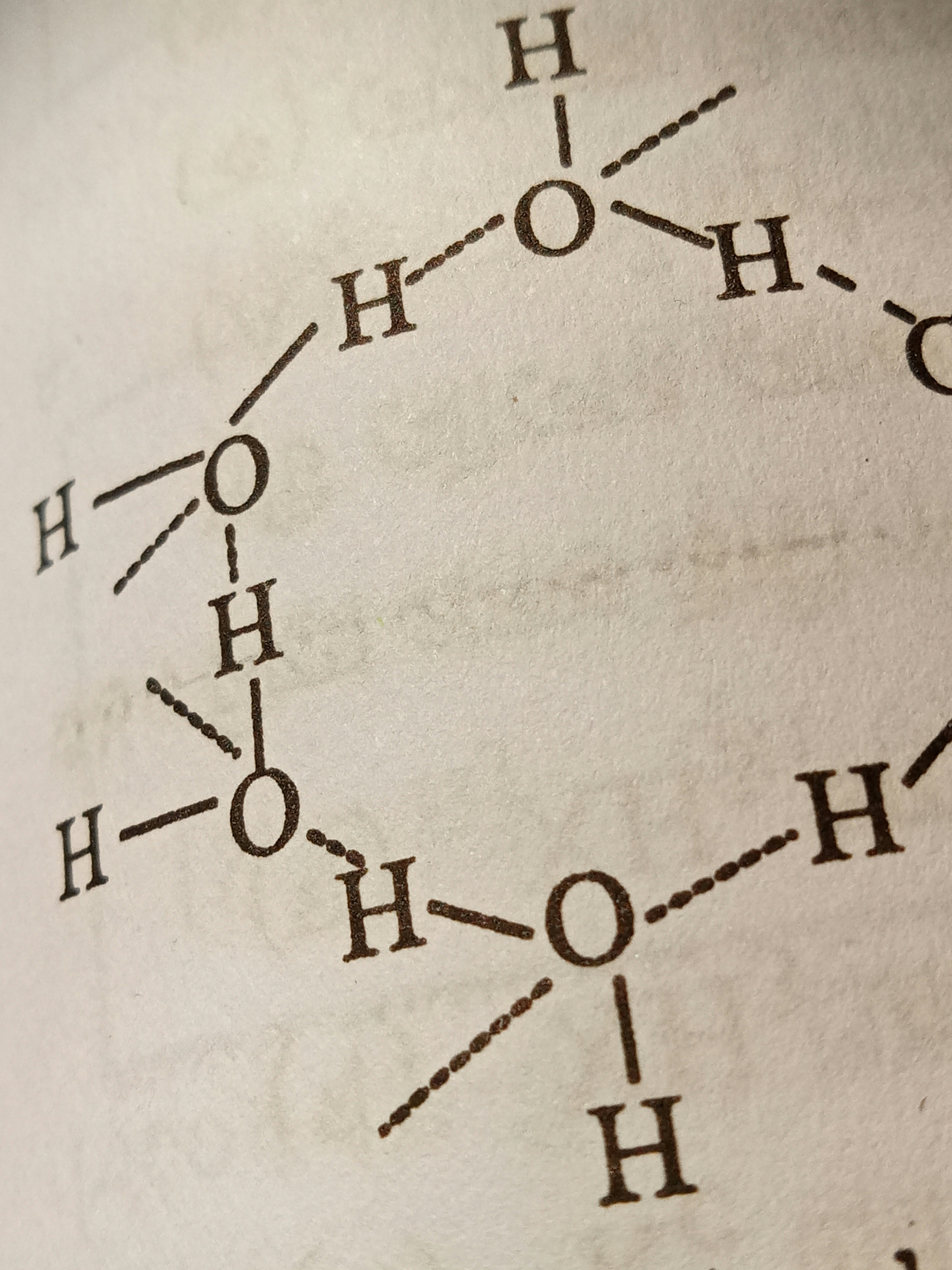
Lastly, hydrogen bonds are a less formal type of interaction that occurs between molecules or within different parts of a single molecule. These bonds form when a hydrogen atom that is covalently bonded to a highly electronegative atom, such as oxygen or nitrogen, experiences attraction to another electronegative atom. Although hydrogen bonds are generally weaker than ionic and covalent bonds, they significantly influence water’s properties and the three-dimensional structures of proteins and nucleic acids.
In summary, understanding the different types of chemical bonds is essential for comprehending how molecules are formed and function within living organisms. The unique characteristics of ionic, covalent, and hydrogen bonds contribute to the diverse array of biological processes that sustain life.
Simple Molecules in Living Organisms
Simple molecules are fundamental components of living organisms, providing the necessary building blocks for more complex biological structures. Among the most vital simple molecules are water, carbon dioxide, and oxygen. These molecules play crucial roles in various biological processes, including cellular respiration, photosynthesis, and metabolic functions.
Water is perhaps the most critical simple molecule for life. Its ability to form hydrogen bonds gives it unique properties, such as high specific heat capacity and solvent capabilities. This makes water an excellent medium for chemical reactions within cells and organisms. Additionally, it acts as a transport medium, facilitating the movement of nutrients and waste products. In cellular processes, water participates in hydrolysis reactions that help break down larger molecules into usable components.
Next, carbon dioxide (CO2) is essential for photosynthetic organisms. Plants and certain microorganisms utilize this simple molecule during the photosynthetic process, converting carbon dioxide and water into glucose and oxygen, using sunlight as a catalyst. This reaction not only sustains the life of photosynthetic organisms but also contributes to the atmospheric balance of gases, making oxygen available for other living beings.
Oxygen (O2), a product of photosynthesis, is equally vital for aerobic organisms. It serves as the final electron acceptor in cellular respiration, a process that extracts energy from glucose for cellular activities. The interaction between these simple molecules—water, carbon dioxide, and oxygen—creates a dynamic environment that supports life. Their role as building blocks for complex biological structures underlines the intricate connections and dependencies among molecules in living organisms.
Complex Molecules and Macromolecules
Complex molecules, particularly macromolecules such as proteins, nucleic acids, carbohydrates, and lipids, are fundamental to the structure and function of living organisms. These macromolecules are large, complex structures that play pivotal roles in biological processes and are synthesized through polymerization, which involves the joining of smaller units known as monomers.
Proteins are composed of amino acids linked by peptide bonds, forming polypeptides that fold into specific three-dimensional shapes. This structure is crucial for their function, as proteins act as enzymes, hormones, structural components, and signaling molecules. Their diverse roles arise from the variation in amino acid sequences, which influences their shape and function. For example, enzymes speed up biochemical reactions, while antibodies protect against pathogens.
Nucleic acids, including DNA and RNA, are polymers made up of nucleotide units. DNA stores genetic information in the sequence of its bases, which determines the synthesis of proteins via RNA. RNA plays an essential role in translating this information into functional proteins, highlighting the intricate relationship between these nucleic acid macromolecules and cellular function. Through processes such as transcription and translation, the information encoded in nucleic acids is expressed into the phenotypic traits of an organism.
Carbohydrates are essential macromolecules composed of sugar monomers that serve as energy sources and structural components. They are classified into monosaccharides, disaccharides, and polysaccharides, with each type serving unique roles from energy metabolism to forming the structural framework of cells, as seen in cellulose and chitin. The complexity of carbohydrates plays a significant role in cell communication and recognition.
Lipids, although not polymers, are vital macromolecules characterized by their hydrophobic nature. They include triglycerides, phospholipids, and steroids. Lipids function in energy storage, form cellular membranes, and serve as signaling molecules. The structure of phospholipids is particularly important in forming bilayers that make up cell membranes, facilitating compartmentalization within cells.
The Role of Bonding in Molecular Shape and Function
Bonding plays a crucial role in determining the shape and overall function of molecules, particularly in biological systems. The type of chemical bond—be it ionic, covalent, or hydrogen—defines not only the internal configuration of the molecules but also their interactions with other molecules. The three-dimensional structure that arises from these bonds significantly influences molecular capabilities, which is particularly evident in enzymes and antibodies.
Covalent bonds are essential for forming stable molecules with specific geometries. For example, the tetrahedral shape of methane (CH4) is a product of covalent bonding, which arises from the arrangement of electron pairs around the central carbon atom. This geometric shape affects how methane interacts with other molecules, including its reactivity and solubility. In enzymes, the particular arrangement of atoms within the active site is optimized for substrate binding, directly impacting the enzyme’s catalytic ability. If the shape of the enzyme were altered due to different bonding interactions, the enzyme might become ineffective, highlighting the importance of bonding in biological function.
Additionally, the role of hydrogen bonding cannot be underestimated, particularly in maintaining the secondary and tertiary structures of proteins. The intricate folding patterns of proteins, essential for their functionality, arise from both covalent and non-covalent bonds, with hydrogen bonds contributing to the formation of alpha-helices and beta-sheets. Antibodies, which are specialized proteins, rely on precise binding shapes to recognize and neutralize pathogens. Any change in the shape caused by variations in bonding would affect their ability to perform this critical function.
Thus, the nature and type of bonding within a molecule are not merely structural aspects; they are fundamental determinants that shape molecular interactions and biological functionalities. The relationship between bonding, molecular shape, and biological function is paramount, demonstrating that even slight variations can lead to significant consequences in living systems.
Interactions Among Molecules: Cohesion and Adhesion
The interactions among molecules play a crucial role in the behavior of living systems, particularly through the phenomena of cohesion and adhesion. Cohesion refers to the attractive forces between molecules of the same substance, which is prominently observed in water. The hydrogen bonds that form between water molecules result in a high degree of cohesion, enabling water to exhibit surface tension. This property allows certain organisms, such as water striders, to walk on water, demonstrating the significance of molecular interactions in their survival.
Adhesion, on the other hand, occurs when molecules of different substances attract each other. This is particularly evident when water molecules adhere to surfaces, such as plant cells. This property is vital for plant life as it aids in capillary action, enabling water to move against gravity from the roots to the leaves through narrow xylem vessels. The combination of cohesion and adhesion is fundamental to the transport of nutrients and water in plants, highlighting the importance of these molecular interactions in biological systems.
Moreover, these properties of water are not just limited to plant life. In animal systems, adhesion allows for the efficient functioning of cells and tissues. For instance, the adhesion of water molecules to the surfaces within biological membranes assists in maintaining cellular structure and function. Similarly, the cohesive forces help to regulate body temperature and facilitate various metabolic processes. Understanding how cohesion and adhesion impact living matter is essential for comprehending the broader implications for life on Earth.
Chemical Reactions and Metabolism
Chemical reactions are fundamental processes that enable living organisms to maintain life. At the core of these processes is metabolism, which encompasses all the biochemical reactions that occur within a cell or an organism. Metabolism is broadly divided into two categories: catabolism, which involves the breakdown of molecules to release energy, and anabolism, where energy is utilized to synthesize complex molecules from simpler ones. Both types of reactions are essential for growth, repair, and overall cellular function.
Enzymes play a crucial role in mediating these chemical reactions. They act as biological catalysts that lower the activation energy needed for reactions to occur, thus increasing the rate of metabolism. Each enzyme is specific to a particular substrate, which is the molecule upon which the enzyme acts. The interaction between enzymes and substrates forms the basis of many metabolic pathways, allowing for the efficient transformation and utilization of energy. This specificity and efficiency are vital for maintaining homeostasis within an organism.
The significance of chemical bonding is evident in the formation of biomolecules such as carbohydrates, proteins, lipids, and nucleic acids. These molecules are produced through various metabolic pathways and serve critical roles in cellular structure and function. For instance, proteins, formed by peptide bonds between amino acids, carry out most cellular functions, including acting as enzymes, signaling molecules, and structural components. The energy transfer that occurs during these reactions often involves high-energy compounds like adenosine triphosphate (ATP), which serves as the primary energy currency within cells.
In summary, chemical reactions are at the heart of metabolic processes in living organisms. The intricate interplay between enzymes, chemical bonds, and energy transfer underscores the complexity of metabolic pathways, enabling life to flourish in myriad forms. Understanding these reactions provides essential insight into the biochemical foundation of life itself.
Conclusion: The Complexity of Living Matter
Throughout this discussion, we have explored the intricate nature of living matter molecules and the complex bonding patterns that underpin the very fabric of life. From the simplest molecular formations to the more intricate structures that define biological organisms, it is evident that these bonds are not mere connections; they are essential interactions that facilitate the myriad functions necessary for life itself.
The complexity of these molecular bonds—be they covalent, ionic, or hydrogen—highlights the sophisticated mechanisms that enable cellular processes such as metabolism, replication, and communication. Living matter is characterized by an astounding array of molecular interactions, each playing a crucial role in maintaining homeostasis and supporting life’s diversity. As we delve deeper into the discipline of molecular biology, we recognize that our understanding of these complex interactions continues to evolve, revealing new dimensions of how molecules operate within living systems.
Moreover, ongoing research emphasizes the necessity of comprehending not only the individual components of living matter but also the dynamic relationships between them. Such an approach is essential for grasping how cellular behaviors arise from molecular interactions, and it can provide insights into the mechanisms that drive various biological phenomena. Thus, the exploration of living matter molecules not only enhances our fundamental knowledge but also opens avenues for medical advancements and biotechnological innovations.
In summary, the intricate nature of living matter molecules challenges us to continue our investigation into their complexities. As we gain a deeper appreciation for the sophisticated bonding patterns that sustain life, we also broaden our capacity to harness this knowledge for the betterment of humanity and the environment.